 您的购物车当前为空
您的购物车当前为空
Ritlecitinib
Synonyms: PF-06651600Ritlecitinib (PF-06651600) 是一种口服有效的选择性JAK3抑制剂,IC50值为 33.1 nM,不影响JAK1/2的活性。
Ritlecitinib
一键复制产品信息别名 PF-06651600
Ritlecitinib (PF-06651600) 是一种口服有效的选择性JAK3抑制剂,IC50值为 33.1 nM,不影响JAK1/2的活性。
| 规格 | 价格 | 库存 | 数量 |
|---|---|---|---|
| 2 mg | ¥ 297 | 现货 | |
| 5 mg | ¥ 483 | 现货 | |
| 10 mg | ¥ 778 | 现货 | |
| 25 mg | ¥ 1,560 | 现货 | |
| 50 mg | ¥ 2,480 | 现货 | |
| 100 mg | ¥ 3,690 | 现货 | |
| 200 mg | ¥ 5,230 | 现货 | |
| 500 mg | ¥ 7,930 | 现货 | |
| 1 mL x 10 mM (in DMSO) | ¥ 535 | 现货 |
TargetMol的所有产品仅用作科学研究或药证申报,不能被用于人体,我们不向个人提供产品和服务。请您遵守承诺用途,不得违反法律法规规定用于任何其他用途。
凭借在化合物合成方面的丰富经验,我们可以根据您的研究需求为该产品提供快速定制合成服务。
产品介绍
| 产品描述 | Ritlecitinib (PF-06651600) is an orally available, selective JAK3 inhibitor with an IC50 of 33.1 nM and does not affect the activity of JAK1/2. |
| 靶点活性 | JAK3:33.1 nM (cell free) |
| 体外活性 | 方法:在体外生理上相关的ATP浓度(1mM)存在的情况下,研究Ritlecitinib对JAK1/2/3激酶的活性影响。 |
| 体内活性 | 方法:Ritlecitinib (PF-06651600) (3,10,30 mg/kg,口服,每天一次)治疗关节炎和脑脊髓炎两种啮齿动物模型小鼠,研究对其影响。 |
| 激酶实验 | His-tagged recombinant human TYK2 kinase domain was expressed in SF21/baculovirus and purified using a two-step affinity (Ni-NTA) and size-exclusion (SEC S200) purification method.Test compounds were solubilized in DMSO to a stock concentration of 30 mM.Compounds were diluted in DMSO to create an 11-point half log dilution series with a top concentration of 600 μM.The test compound plate also contained positive control wells containing a known inhibitor to define 100% inhibition and negative control wells containing DMSO to define no inhibition.The compound plates were diluted 1 to 60 in the assay,resulting in a final assay compound concentration range of 10 μM to 100 pM and a final assay concentration of 1.7% DMSO.Test compounds and controls solubilized in 100% DMSO were added (250 nL) to a 384 well polypropylene plate (Matrical) using a non contact acoustic dispenser.Kinase assays were carried out at room temperature in a 15 μL reaction buffer containing 20 mM HEPES,pH 7.4,10 mM magnesium chloride,0.01% bovine serum albumin (BSA),0.0005% Tween 20 and 1mM Dithiothreitol (DTT).Reaction mixtures contained 1 μM of a fluorescently labeled synthetic peptide,at a concentration less than the apparent Michaelis-Menten constant (Km) (5FAM-KKSRGDYMTMQID for JAK1 and TYK2 and FITC-KGGEEEEYFELVKK for JAK2 and JAK3).Reaction mixtures contained adenosine triphosphate (ATP) at either a level equal to the apparent Km for ATP (40 μM for JAK1,4 μM for JAK2,4 μM for JAK3 and 12 μM for TYK2) or at 1 mM ATP.Compound was added to the buffer containing ATP and substrate and immediately after this step the enzyme was added to begin the reaction.The assays were stopped with 15 μL of a buffer containing 180 mM HEPES,pH=7.4,20 mM EDTA,0.2% Coating Reagent,resulting in a final concentration of 10 mM EDTA,0.1% Coating Reagent and 100 mM HEPES,pH=7.4. |
| 细胞实验 | Human CD4+ T cells were purified from buffy coat with RosetteSep CD4+ T Cell Enrichment Cocktail and skewed for 6 days with cytokine cocktails (25 ng/mL of IL-6, 25 ng/mL of IL-23, 12.5 ng/mL of IL-1β, 25 ng/mL of IL21, 5 ng/mL of TGFβ1, 10 μg/ml of anti-CD3 antibody (pre-coated on plate surface) and 1 μg/mL of anti-CD28 antibody) in the presence of JAK inhibitors at 10 different concentrations. Supernatants were harvested and the concentrations of IL-17A were determined with MSD assay following the protocol provided by the manufacturer. To study the effect of PF-06651600 on Th17 cells post-differentiation, skewed Th17 cells were washed, rested with X-VIVO 15 medium for overnight and resuspended in medium containing the same concentrations of cytokines as during skewing but without anti-CD3 or anti-CD28 antibodies, in the presence of PF-06651600 at 10 different concentrations for 2 additional days. On Day 9, supernatant was harvested from each well and IL-17A was determined as described above [1]. |
| 动物实验 | The effect of JAK3 inhibition by PF-06651600 was evaluated in vivo using a therapeutic dosing paradigm in a rat adjuvant-induced arthritis. The efficacy of this molecule was evaluated in three separate studies using successively lower doses. Arthritis was induced by immunization of female Lewis rats (8 to 10 weeks old) via intradermal injection at the base of the tail with complete Freund's adjuvant with three 50 μL injections (15 mg/mL Mycobacterium tuberculosis) in incomplete Freund's adjuvant. Seven days after the initial immunization, the baseline hind paw volume of the immunized rats was measured via plethysmograph. The rats were monitored daily for signs of arthritis including change in body weight and hind paw volume measurement. When individual hind paw volume measurements indicated an increase of 0.2 mL (or greater) in a single hind paw, animals were randomly assigned to a treatment group. Daily treatment with PF-06651600 was administered via oral gavage. Treatment groups for Experiment 1 were: 80, 15, or 6 mg/kg or vehicle (2% Tween 80 /0.5% methylcellulose/deionized water). Treatment groups for Experiment 2 were: 30, 10, and 3 mg/kg or vehicle (0.5% methylcellulose / de-ionized water/ 1 mEQ hydrochloric acid). Treatment groups for Experiment 3 were: 10, 1, 0.3 and 0.1 mg/kg or vehicle (0.5% methylcellulose/de-ionized water/ 1 mEQ hydrochloric acid). Dosing began once individuals were enrolled into respective groups. Treatment continued for 7 days. At the conclusion of the study, whole blood was taken at 15 minutes post-dose (peak concentration in plasma) for analysis of STAT phosphorylation, and plasma was taken for exposure concentration in PF-06651600 dosed groups [1]. |
| 别名 | PF-06651600 |
| 分子量 | 285.34 |
| 分子式 | C15H19N5O |
| CAS No. | 1792180-81-4 |
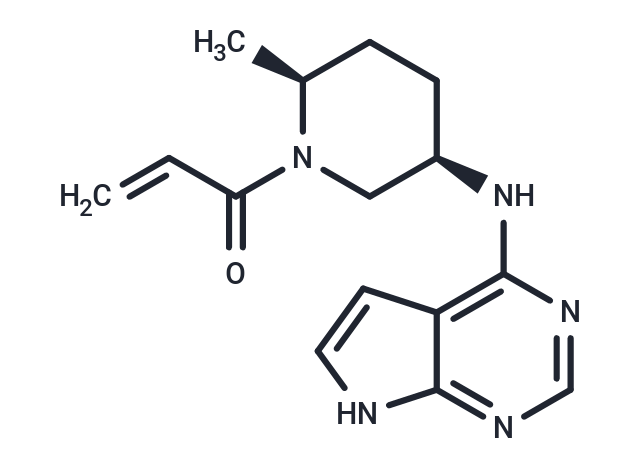
| Smiles | C[C@H]1CC[C@H](CN1C(=O)C=C)Nc1ncnc2[nH]ccc12 |
| 密度 | 1.272 g/cm3 (Predicted) |
| 存储 | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. 实际储存温度请以COA为准 | |||||||||||||||||||||||||||||||||||
| 溶解度信息 | DMSO: 130 mg/mL (455.6 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
| 体内实验配方 | 10% DMSO+40% PEG300+5% Tween 80+45% Saline: 4 mg/mL (14.02 mM), Sonication is recommended. 请按顺序添加溶剂,在添加下一种溶剂之前,尽可能使溶液澄清。如有必要,可通过加热、超声、涡旋处理进行溶解。工作液建议现配现用。以上配方仅供参考,体内配方并不是绝对的,请根据不同情况进行调整。 | |||||||||||||||||||||||||||||||||||
溶液配制表 | ||||||||||||||||||||||||||||||||||||
DMSO
该溶液配制表仅适用于固体产品。对于液体产品,请根据标明的浓度或密度计算稀释方案。 | ||||||||||||||||||||||||||||||||||||
计算器
体内实验配液计算器
剂量转换
对于不同动物的给药剂量换算,您也可以参考 更多




 很棒
很棒
 |
|